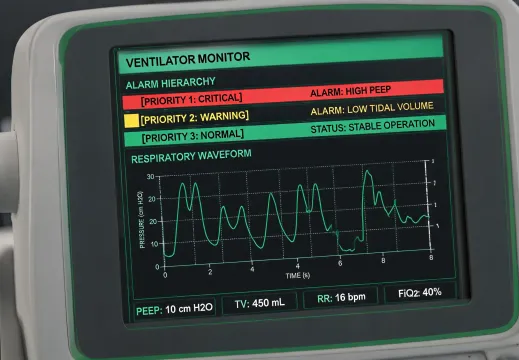
Readable alarm hierarchy
High, medium, and low-priority alarms need visible contrast, concise wording, and staff education that explains what to do next. A good interface reduces hesitation without hiding clinically important warnings.
Hamilton Medical uses design reviews to make clinical work calmer: clear labels, visible alarm states, easier cleaning, fewer hidden consumable steps, and documentation that helps staff trust the device before the first patient encounter.

High, medium, and low-priority alarms need visible contrast, concise wording, and staff education that explains what to do next. A good interface reduces hesitation without hiding clinically important warnings.
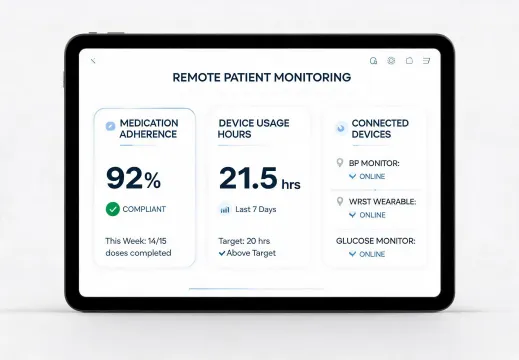
Home-health teams need adherence, usage hours, connection state, and caregiver notes in one view. The design goal is not decoration; it is faster triage with fewer avoidable calls.
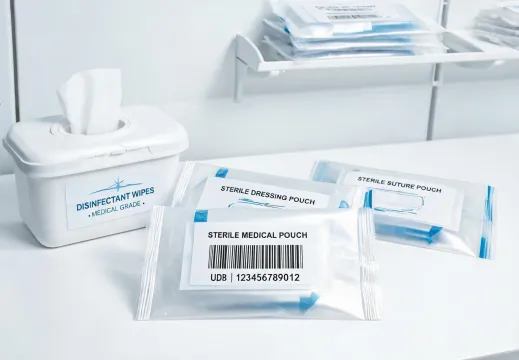
Packaging, UDI labels, IFU access, and replenishment cues help infection-control products move through a facility without losing lot visibility or confusing staff during a busy shift.

Field engineers work better when critical tools, spare parts, checklists, and service notes are physically and digitally organized around the task they are about to perform.
Clinical users should not decode a device in stressful moments. Labels, alerts, and quick guides should point to the next responsible action.
Surfaces, packaging, barcodes, and instructions should support infection-control routines and lot visibility without adding unnecessary steps.
Respiratory therapists, nurses, biomed engineers, supply chain teams, and caregivers all see different parts of the same workflow. Good design speaks to each of them.
This gallery is intentionally practical. It does not claim that visual polish alone improves outcomes. Instead, it shows how thoughtful design can reduce avoidable confusion, support training, improve service readiness, and help regulated documentation remain close to daily clinical work.
Share a device workflow, training challenge, or consumables process, and a Hamilton Medical advisor will suggest where design changes may reduce friction.
Request Design Guidance